The carcinogenic potential of nitrosamines has been known for a long time. Nitrosamines such as N-nitrosodimethylamine (NDMA) are metabolically degraded to alkylating reagents that can methylate DNA bases(1). The compounds are mainly found in smoked or cured meat and fish products, in tobacco smoke, but also in cosmetics.
Incidentally, several human drugs have also come into focus as nitrosamine sources due to impurities caused by the starting materials used for the synthesis of active ingredients. For example, nitrosamines have been found as impurities in known antihypertensives(2), gastric acid blockers(3), and antidiabetics(4).
Many reactants in synthesis contain secondary or tertiary amino functional groups that can react with nitrous acid or other nitrosating reagents to form nitrosamines. Solvents, such as DMF, can also form nitrosamines under certain reaction conditions with secondary amines.
Another potential source of nitrosamine impurities is through the potable water used in manufacturing the drug API. It is therefore the responsibility of the manufacturers to regularly check the water quality for nitrite and nitrosamine limits(5).
Nitrosamines do not always have to be supplied 'exogenously', but can also be formed 'endogenously' in human metabolism by reaction with saliva or gastric juice(6). Therefore, the "nitrosamine formation potential" from nitrosatable amines must also be considered one’s risk assessment.
N-nitrosodimethylamine (NDMA)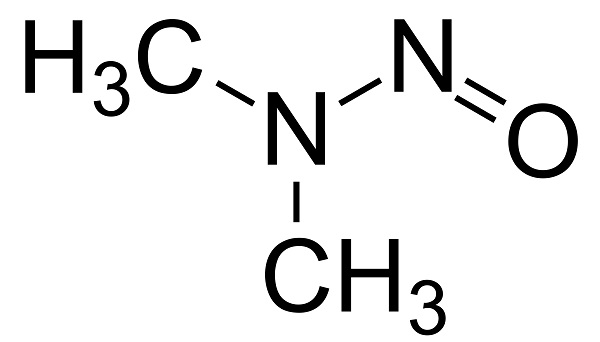
Today, nitrosamines are determined by mass spectrometry coupled with HPLC or GC. The following GC/MS analysis of four nitrosamines was performed in the application laboratory of MACHEREY-NAGEL's trading partner Beijing Zhenxiang Technology. MACHEREY-NAGEL’s low bleed 5% diphenyl 95% dimethylpolysiloxane OPTIMA 5 MS capillary column was utilized.
Access to application: Water quality - determination of nitrosamine compounds by gas chromatography
References:
(1) P.F. Swann and P.N. Magee, Biochem. J. (1968) 110,39.
(2) "FDA Updates and Press Announcements on Angiotensin II Receptor Blocker (ARB) Recalls (Valsartan, Losartan, and Irbesartan)". Drug Safety and Availability. U.S. Food and Drug Administration (FDA). November 7, 2019. Retrieved November 15, 2019.
(3) "Statement alerting patients and health care professionals of NDMA found in samples of ranitidine". U.S. Food and Drug Administration (FDA). 13 September 2019. Archived from the original on 26 September 2019. Retrieved 26 September 2019. This article incorporates text from this source, which is in the public domain.
(4) "FDA Updates and Press Announcements on NDMA in Metformin". U.S. Food and Drug Administration (FDA). 2 July 2020. Retrieved 11 July 2020.
(5) See latest version of the WHO’s Guidelines for Drinking-Water Quality at https://www.who.int/water_sanitation_health/water-quality/guidelines/en/.
(6) Labor Veritas Spotlight, May 2020, (German), www.laborveritas.


